

1 / 5










| Customization: | Available |
|---|---|
| Application: | Orthopedic, Neurosurgery, Microsurgery |
| Material: | Steel |
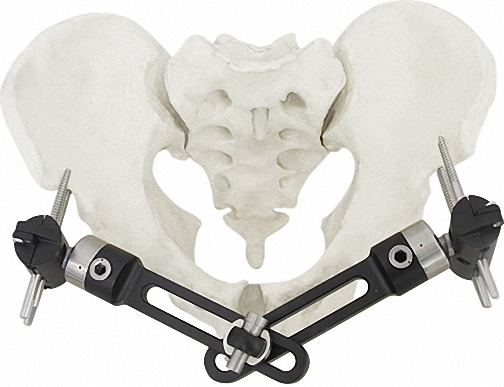
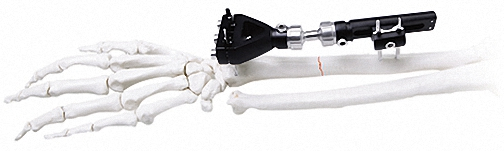
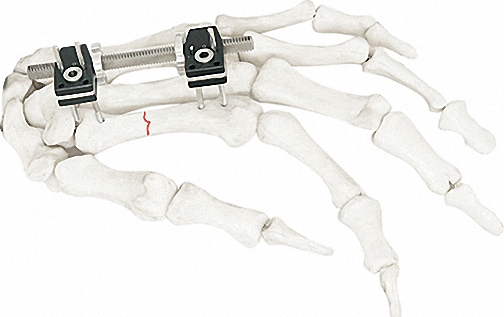
The Pinnacle of Modular Fixation Systems



| Component Description | Specification | Quantity |
|---|---|---|
| Independent Coupling | φ6*φ14 (1.5-2.0) | 10 |
| Needle Clip | φ4*φ9 (φ1.0) | 5 |
| Cone Washer | φ4*φ9 | 10 |
| Double Groove Needle Clamp | φ4*φ14(2.0-2.5) | 5 |
| Post With Threaded Post | φ4*15(1 Hole)*M4 | 8 |
| Connection Plate | φ4*20 (2 Hole) | 4 |
| Hex Nut | M3 / M4 | 90 |
| Bone Screws | 1.0*150 / 1.2*150 / 1.5*150 | 20/20/8 |
| Instrument Set | Standard Configuration | 1 |

Lower Limbs Lengthening External Fixator

Orthopedic External Fixator (Humeral & Tibial)

Ring Fixator for Tibia Femur

2/3 Ring Fixator

Ankle Joint Fixator

Knee Joint Fixator
 DTRX Orthopedic Implants
DTRX Orthopedic Implants