Product Description
Application: Widely used for different bone fracture of different bones.
Available Head: Hexagonal head.
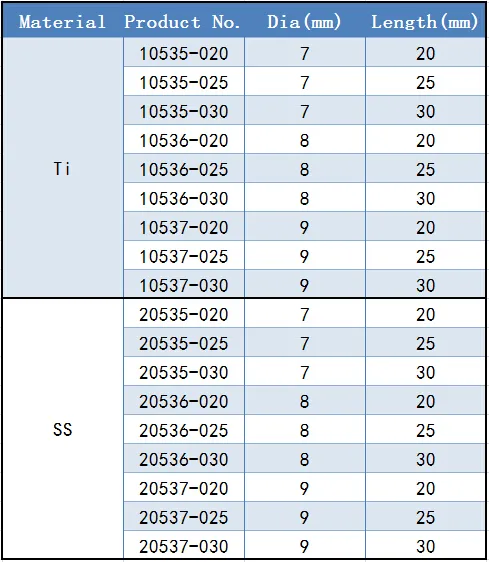
Available Diameter & Length:
• 7.0mm: 20/25/30mm
• 8.0mm: 20/25/30mm
• 9.0mm: 20/25/30mm
Instrument: ACL Interference Screw Instruments.
Technical Excellence
As a national high-tech enterprise, the R&D investment consistently exceeds 8% of total sales. This commitment facilitates the transformation of clinical expertise into innovative medical achievements. Key proprietary technologies include the Rev Drill system, Three-Row-Stapler, Sternal Fixation, and Rib plates, which are recognized benchmarks in the domestic medical devices industry.
Manufacture & Quality Control
Production is conducted strictly under the ISO13485 quality system and CE MDD 93/42/EEC directive. Our facilities utilize advanced German-imported and American-imported machining centers, along with Japanese-imported automatic lathes, to ensure first-class manufacturing precision.
The dedicated Testing Center comprises a biomechanical laboratory and a physical-chemical testing center. These facilities provide rigorous assurance across three critical stages: raw material procurement control, product quality control, and new product performance analysis.
Global Presence
Our surgical products are exported globally to countries including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. Feedback from international markets confirms that our product quality consistently meets local regulatory standards.
Frequently Asked Questions
What materials are the interference screws made of?
The screws are available in Pure Titanium, Titanium Alloy, and Stainless Steel options to suit different clinical requirements.
What diameters and lengths are available?
We provide diameters of 7.0mm, 8.0mm, and 9.0mm, with standard lengths of 20mm, 25mm, and 30mm.
Are these products certified for international use?
Yes, the products are certified under CE, ISO13485, and SGS standards, making them suitable for global medical markets.
How is the quality of the raw materials ensured?
We maintain a physical-chemical testing center specifically for raw material procurement control to ensure all materials meet strict medical grade specifications.
What type of packaging is used for transport?
The products are typically provided in non-sterile packaging using paper-plastic roll bags, designed for safe international transport.
What instruments are required for the procedure?
These screws are designed to be used with specific ACL Interference Screw Instruments for optimal surgical outcomes.

















 DTRX Orthopedic Implants
DTRX Orthopedic Implants