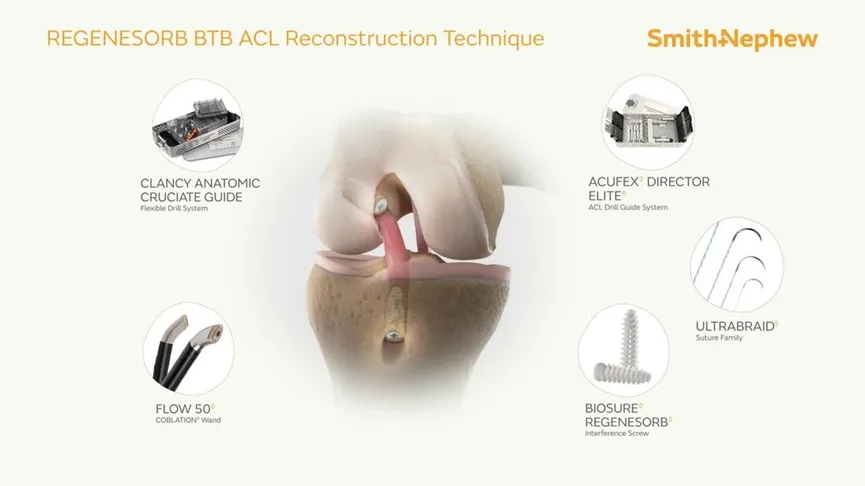
The Interface Screw is designed for use in ACL reconstruction surgeries to secure tendon or ligament grafts in the femoral and tibial bone tunnels. It provides a strong and reliable fixation by compressing the tendon or ligament graft directly against the bone tunnel wall. The screw's unique mechanism ensures optimal stability and a firm attachment between the soft tissue graft and the bone.
The Interface Screw system includes a conical screw, which facilitates easy insertion, and a protective sheath that shields the soft tissue during insertion and reduces the risk of graft displacement or screw breakage. This system is made from PEEK (Polyetheretherketone), a biocompatible material that mimics the elasticity of natural bone, ensuring superior performance during the healing process.
Conical Screw Design
The screw's tapered shape ensures easy insertion, with the head diameter larger than the tip to ease its placement and provide a firm grip.
Star Drive Mechanism
Prevents damage to the screw due to insufficient strength and enhances the holding power, ensuring the screw remains intact under torque.
Protective Sheath
The circular sheath protects the tendon and ligament from being cut by the screw, minimizing the risk of damage during insertion.
Locking Mechanism
Features a clamp block on its outer surface that ensures the graft remains securely in place, even under external pressure.
PEEK Material
Offers excellent biocompatibility and elasticity similar to natural bone, promoting optimal healing and integration of the graft.
This Interface Screw System is primarily used for ACL reconstruction surgeries, where it provides secure fixation for soft tissue grafts or bone-tendon grafts in the knee joint, ensuring effective healing and recovery.
❓ Frequently Asked Questions
What shipping methods are available and what are the costs?
We typically utilize courier services for shipping, but we can also accommodate postal services upon your request. Shipping costs are determined by the destination, package size, and weight, and we will provide a detailed quote once we have your specific requirements.
Are your medical products certified?
Yes, our manufacturing processes hold the EN ISO 13485 certification, which is a testament to our commitment to quality and regulatory compliance in the medical device industry. This ensures products meet stringent requirements for safety and performance.
What is the minimum order quantity (MOQ)?
Our standard minimum order value is US$1000. However, we recognize the need for evaluating products through sample orders. We offer samples at cost upon request, allowing you to test the market before committing to a larger order.
Can we request custom products or items not listed in the catalogue?
Yes, we can accommodate custom orders. Please provide us with pictures or samples of the desired product, and we will work with you to create a similar item. Please note that custom orders may have a longer lead time for delivery.
Do you maintain stock for listed products?
We maintain stock for most of the products listed in our catalogue, with regular monthly production cycles. However, stock levels can vary based on sales and demand. For the most accurate inventory information, it's best to consult with our sales team.











 DTRX Orthopedic Implants
DTRX Orthopedic Implants