



1 / 5










| Still deciding? Get samples of $ ! US$ 100/Piece Request Sample |
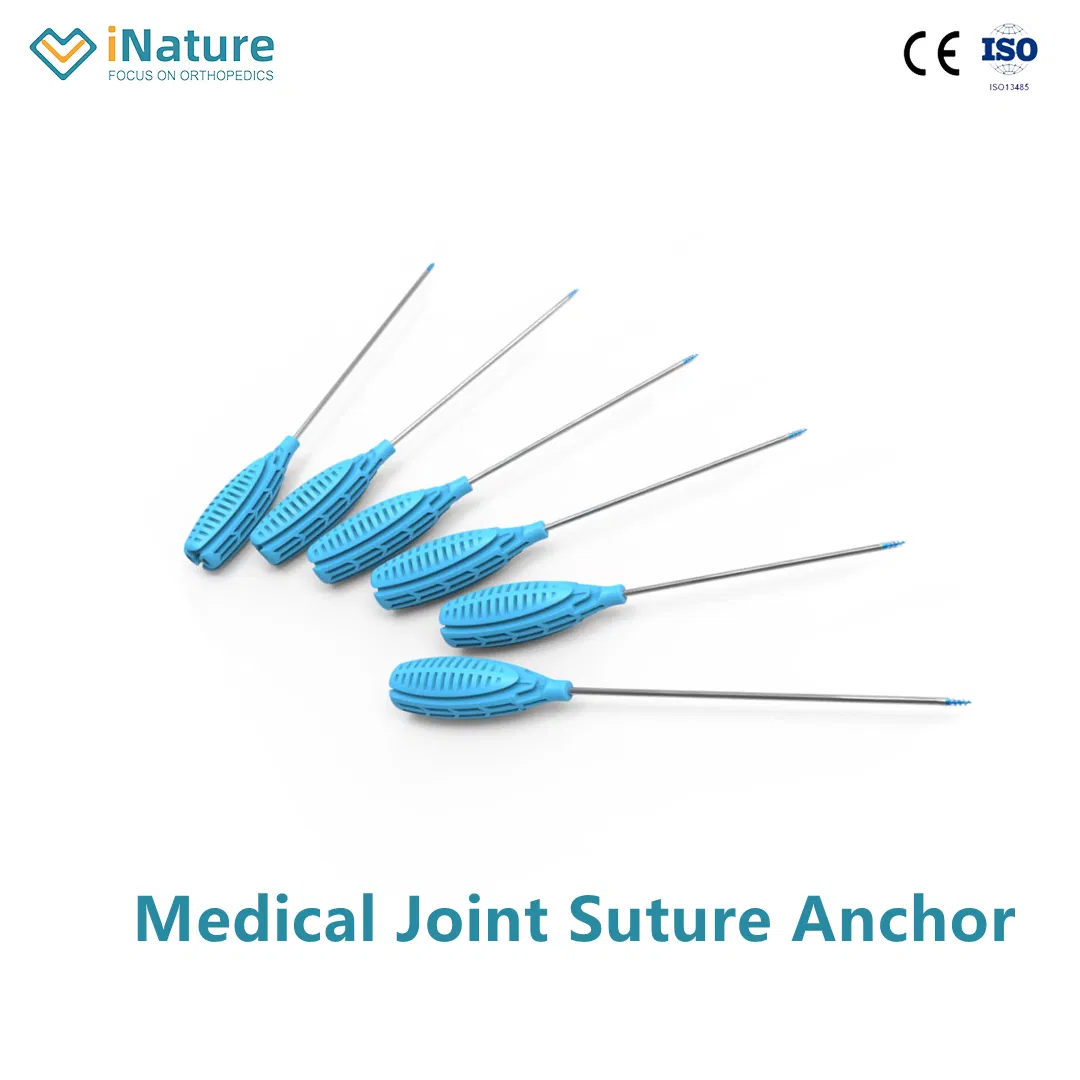
Suture anchors are miniature internal fixation implants widely used in orthopedics, sports medicine, and soft tissue repair surgery. Their core function is to reliably fix sutures to the bone, thereby pulling torn tendons, ligaments, glenoid labrum, and other soft tissues back and fixing them to their anatomical insertions, promoting healing.


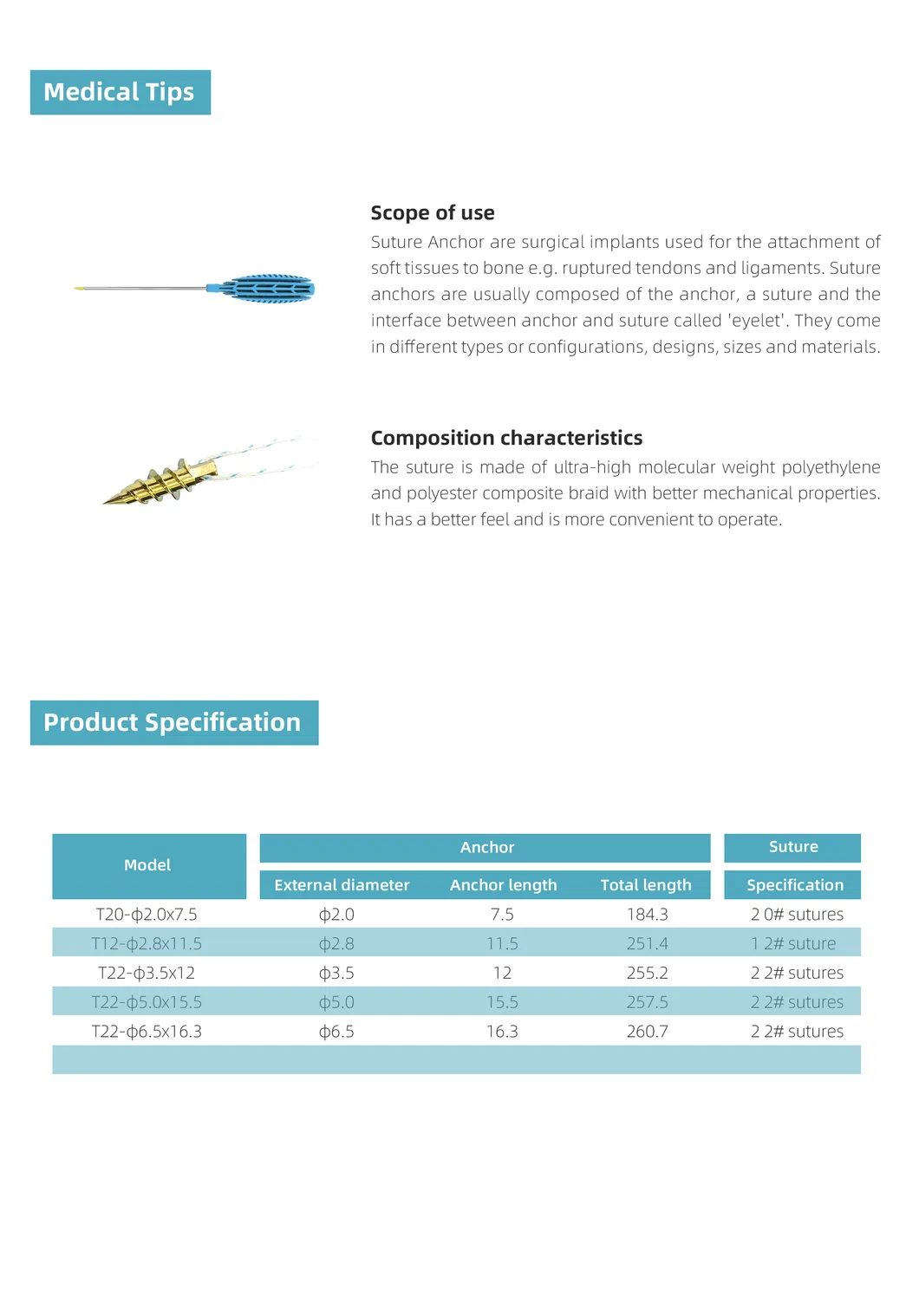
| Product Name | Specification | Anchor Outer Diameter (mm) | Anchor Length (mm) | Total Length (mm) | Suture Specification |
|---|---|---|---|---|---|
| Suture Anchor For Joint Tendon Repair | T12-φ2.0×7.5 | φ2.0 | 7.5 | 184.3 | 2 #0 |
| T12-φ2.8×11.5 | φ2.8 | 11.5 | 251.4 | 1 #2 | |
| T12-φ3.5×12 | φ3.5 | 12 | 255.2 | 2 #2 | |
| T12-φ5.0×15.5 | φ5.0 | 15.5 | 257.5 | 2 #2 | |
| T12-φ6.5×16.3 | φ6.5 | 16.3 | 260.7 | 2 #2 |
*All prices are for reference only. Exact price will be based on the latest quotation.




 DTRX Orthopedic Implants
DTRX Orthopedic Implants