













| Still deciding? Get samples of $ ! US$ 15/Piece Request Sample |

Revolutionize spinal surgery with this mature implant system. Designed for precision and reliability, this system offers both sterile and non-sterile packaging options to meet diverse clinical requirements.


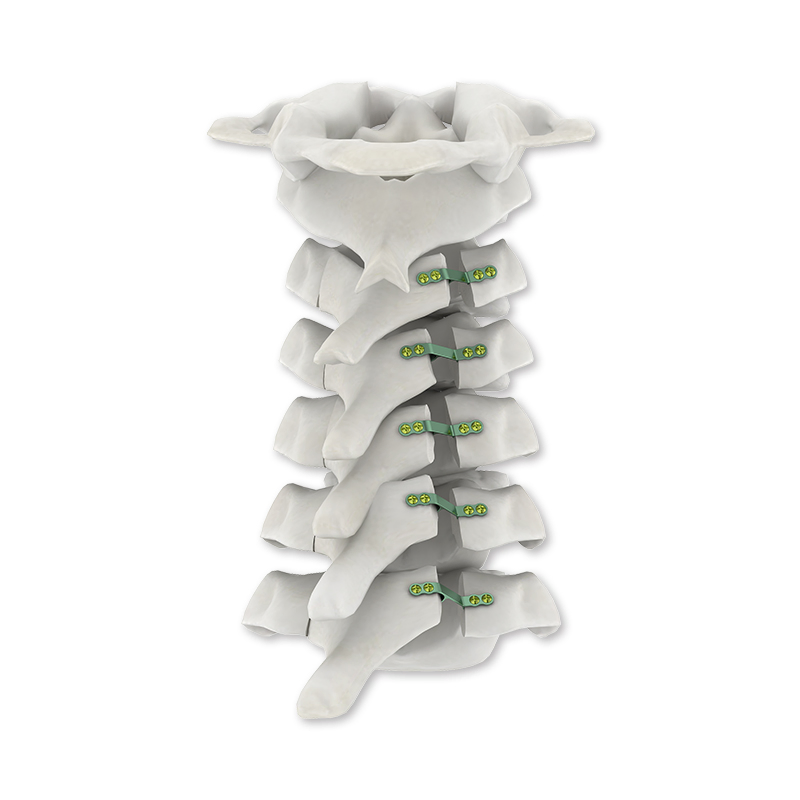
| Product name | Spinal Internal Fixation 5.5/6.0 U-Multi-Axial Reduction Pedicle Screw |
| Material | Titanium |
| Color | Gold, Blue, Grey, Pink, etc |
| Related Rod | 5.5/6.0mm |
| Diameter | 4.5/5.0/5.5/6.0/6.5/7.0 mm |
| Length | 30/35/40/45/50/55/60 mm |
| Certificates | CE/ISO 13485 & 9001 |
| Package | Non-Sterilized / Sterilized |


Our pedicle screw systems are manufactured under strict quality control measures. We offer customizable branding, a variety of color options, and quick sample delivery within 3 days to support clinical evaluation.
The system is primarily constructed from high-grade Titanium, known for its excellent biocompatibility, strength, and durability in spinal fusion procedures.
We offer a wide range of diameters (4.5mm to 7.0mm) and lengths (30mm to 60mm) to accommodate various patient anatomies and surgical requirements.
Yes, we provide both Non-Sterilized and Ethylene Oxide (EO) Sterilized packaging options depending on your facility's needs.
The screws and components are available in multiple colors including Gold, Blue, Grey, and Pink, which can help in distinguishing different sizes or types during surgery.
Our products are fully certified with CE and ISO 13485 & 9001 standards, ensuring they meet international requirements for medical device safety and quality.
Yes, we offer logo printing and custom branding options to meet the specific needs of medical distributors and healthcare providers.
 DTRX Orthopedic Implants
DTRX Orthopedic Implants