














| Still deciding? Get samples of $ ! US$ 40/Piece Request Sample |

| Condition | New |
| Usage | Fracture Fixation |
| Using Site | Bone |
| Type | Internal Fixation |
| Material | Pure Titanium |
| Shape | Bullet Shaped Head |
| Properties | Implant Materials & Artificial Organs |
| OEM | Available |
| Origin | China |
| Production Capacity | 50000 PCS/Year |

| Product Specification | Length (mm) | Material |
|---|---|---|
| φ2.5mm Conical Headless Compression Screw - II | 8/10/12/14/16/18/20/22/24/26/28/30 | Titanium Alloy (TA) |
| φ3.0mm Conical Headless Compression Screw - I | 12 to 40 | Titanium Alloy (TA) |
| φ3.5mm Conical Headless Compression Screw - I | 16 to 45 | Titanium Alloy (TA) |
| φ4.0mm Conical Headless Compression Screw - I | 16 to 50 | Titanium Alloy (TA) |
| φ5.0mm Conical Headless Compression Screw - I | 25 to 80 | Titanium Alloy (TA) |
| φ6.5mm Conical Headless Compression Screw - I | 35 to 120 | Titanium Alloy (TA) |

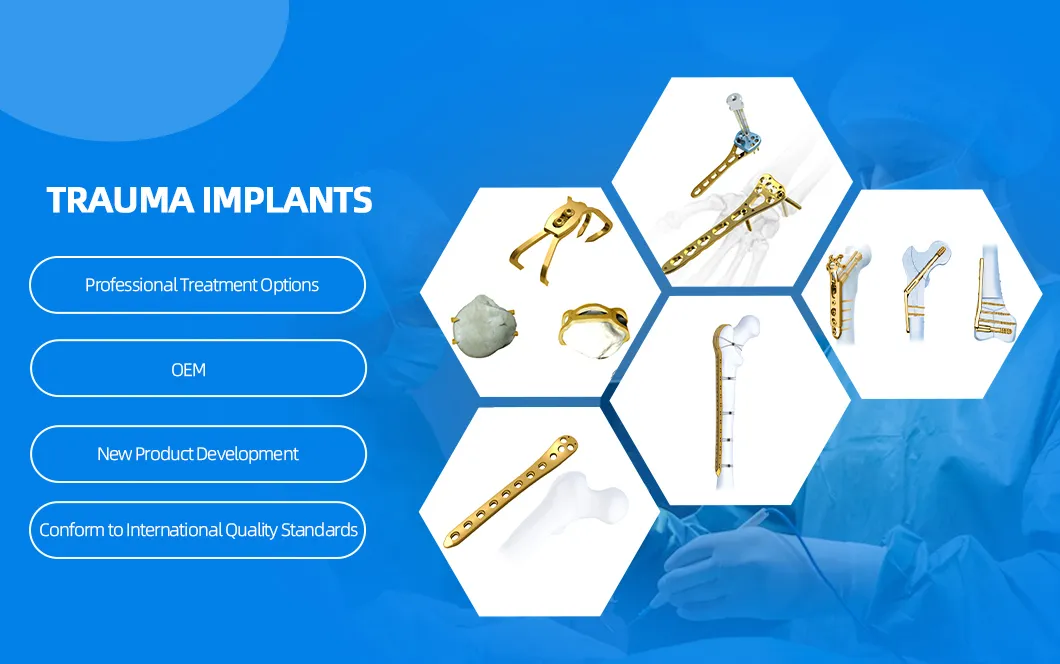
Our company is a leading orthopedic product manufacturer dedicated to providing cost-effective surgical and reliable therapeutic solutions to surgeons and patients worldwide. We offer a comprehensive range of trauma implants, spinal implants, and specialized surgical instruments.
Key categories include locking plates, intramedullary nails, cannulated headless screws, and cable systems for trauma care. For spinal procedures, we provide cervical plates, pedicle screws, and PEEK cages. All products are manufactured under strict quality systems, complying with international standards.




Our products are certified by TUV CE0123, NMPA, and we operate in full compliance with ISO13485 quality management systems to ensure the highest safety standards for clinical use.

 DTRX Orthopedic Implants
DTRX Orthopedic Implants