



1 / 5










| Still deciding? Get samples of $ ! US$ 30/Piece Request Sample |

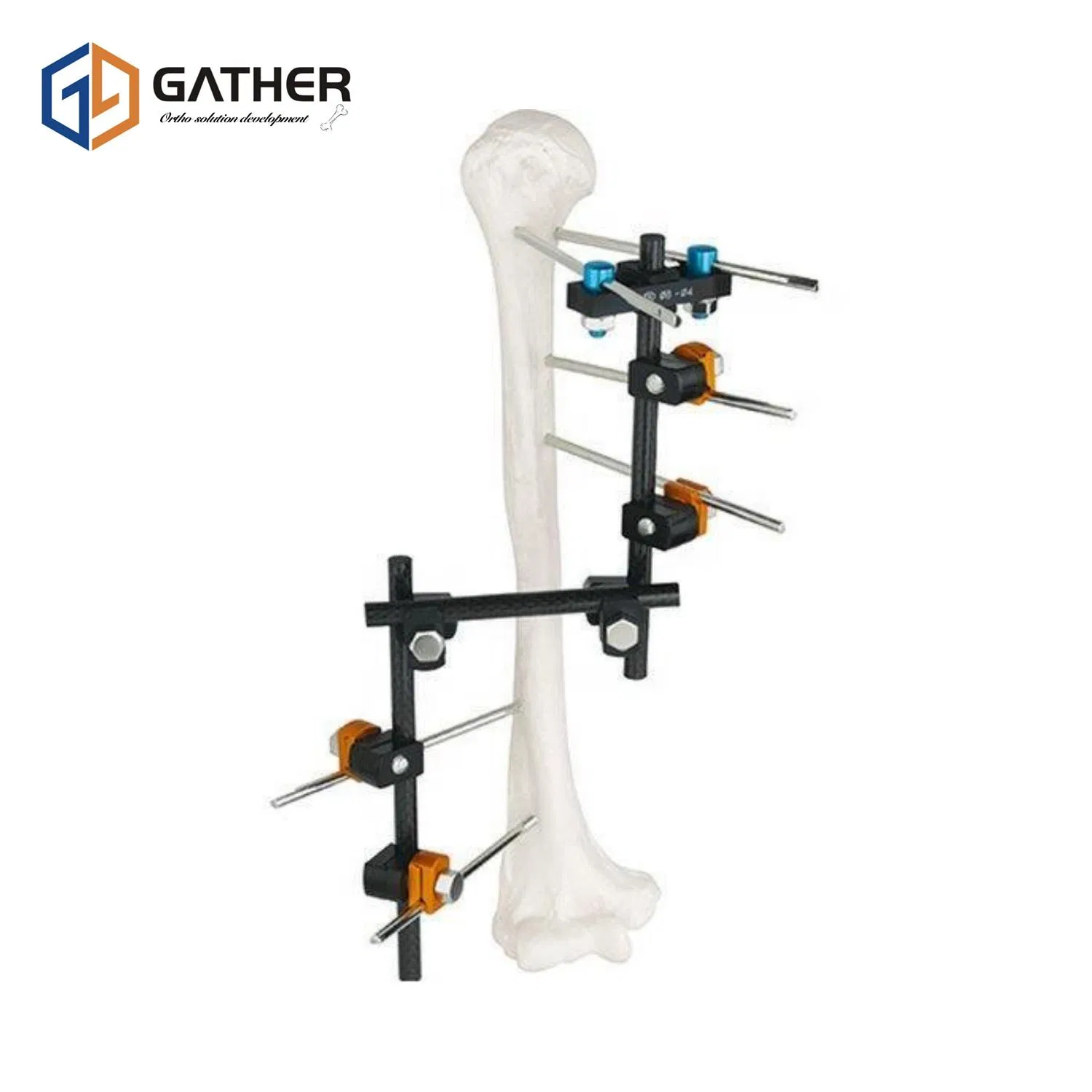
Due to its versatility, the Hoffmann II System is indicated for complete and temporary fracture fixation for Tibia, Femur, Pelvis, and Humerus fractures. It is particularly suited for the following indications:
| Name | Orthopedic External Fixator Rod clamp Upper & Lower Limbs Instruments |
| Material | Stainless Steel |
| Valid Time | 3 Years |
| Licence number | CE/ISO9001 |
| Usage | Medical Fixation Equipment |










 DTRX Orthopedic Implants
DTRX Orthopedic Implants