Basic Information
Model NO.
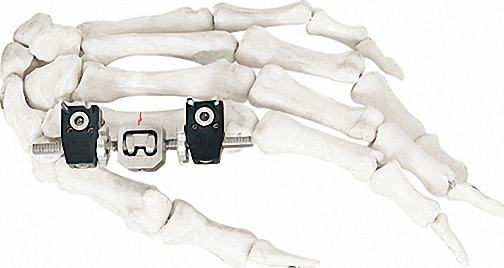
External Fixation
Certification
CE, ISO13485
Material Type
Titanium Alloy & Stainless Steel
Production Capacity
500000 Pieces/Year
Product Description
External Fixator -- The Pinnacle of Modular Fixation Systems: Experience the future of orthopedic care with this revolutionary External Fixator.
◈Distinguished Technology: Revolutionary technology and intuitive, user-friendly operation.
◈Independent Arrangement: Facilitates independent arrangement of bone screws, ideal for proximal and distal fractures.
◈Snap-fit Design: Ensures streamlined and time-efficient surgical procedures.
◈Premium Materials: Enhances fracture visualization with Medical aluminum and carbon fiber.
◈Stability & Flexibility: Provides unparalleled stability catering to diverse medical needs.
◈Compact Frame: Space-saving frame for ease of use and transport.
Technical Component Specifications
| Component Name |
Specification Size |
Quantity |
| Independent Coupling | φ6*φ14(1.5-2.0) / φ6*φ14(1.5) | 10 / 10 |
| Needle Clip | φ4*φ9 (φ1.0) / φ4*φ9 (φ1.5) | 5 / 5 |
| Cone Washer | φ4*φ9 | 10 |
| Double Groove Needle Clamp | φ4*φ14(2.0-2.5) | 5 |
| Cone Washer (Large) | φ4*φ14 | 5 |
| Post With Threaded Post | φ4*15(1 Hole)*M4 / φ4*25(2 Hole)*M4 / φ3*15(1 Hole)*M3 | 8 / 8 / 8 |
| Connection Plate | φ4*20 (2H) to φ4*50 (5H) | 4 per size |
| Straight Connection Plate | φ4 / φ3 | 4 / 4 |
| Fixator (Hinge) | M4*20mm | 4 |
| Straight Threaded Rod | M3 (40-120mm) / M4 (20-155mm) | Various (2-8) |
| Bone Screws | 1.0*150 / 1.2*150 / 1.5*150 | 20 / 20 / 8 |
| Hex Nut | M3 / M4 | 90 / 90 |
| Instrument Set | Standard Case | 1 |
Product Variations & Specific Applications

Lower Limbs Lengthening External Fixator (Tibia/Femur)

Orthopedic Fixator (Humeral & Tibial Lateral Epicondyle)

Ring Fixator (Tibia Femur)

2/3 Ring Fixator (Tibia Femur)

Ankle Joint Fixator (Equinus)

Knee Joint Fixator
Frequently Asked Questions
Q1: What materials are used in the External Fixator?
The system is crafted using high-grade Titanium Alloy, Stainless Steel, Medical Aluminum, and Carbon Fiber to ensure both lightweight handling and maximum stability.
Q2: Is this fixation system reusable?
Yes, the external fixator components are designed to be reusable after proper sterilization, following standard medical protocols.
Q3: Does the system support complex fracture management?
Absolutely. It features independent bone screw arrangement and a modular design specifically engineered for complex fractures and distal/proximal fracture management.
Q4: How does the snap-fit design benefit the surgical process?
The snap-fit design allows for quick assembly and adjustment during surgery, significantly reducing operative time and streamlining the clinical procedure.
Q5: Which certifications does the product hold?
Our External Fixator systems are CE and ISO13485 certified, meeting international standards for medical device safety and quality.
Q6: Can it be used for limb lengthening?
Yes, we provide specific variations such as the Lower Limbs Lengthening External Fixator suitable for both Tibia and Femur procedures.























 DTRX Orthopedic Implants
DTRX Orthopedic Implants