


1 / 5










| Customization: | Available |
|---|---|
| Certification: | FDA, ISO, CE |
| Disinfection: | Without Disinfection |

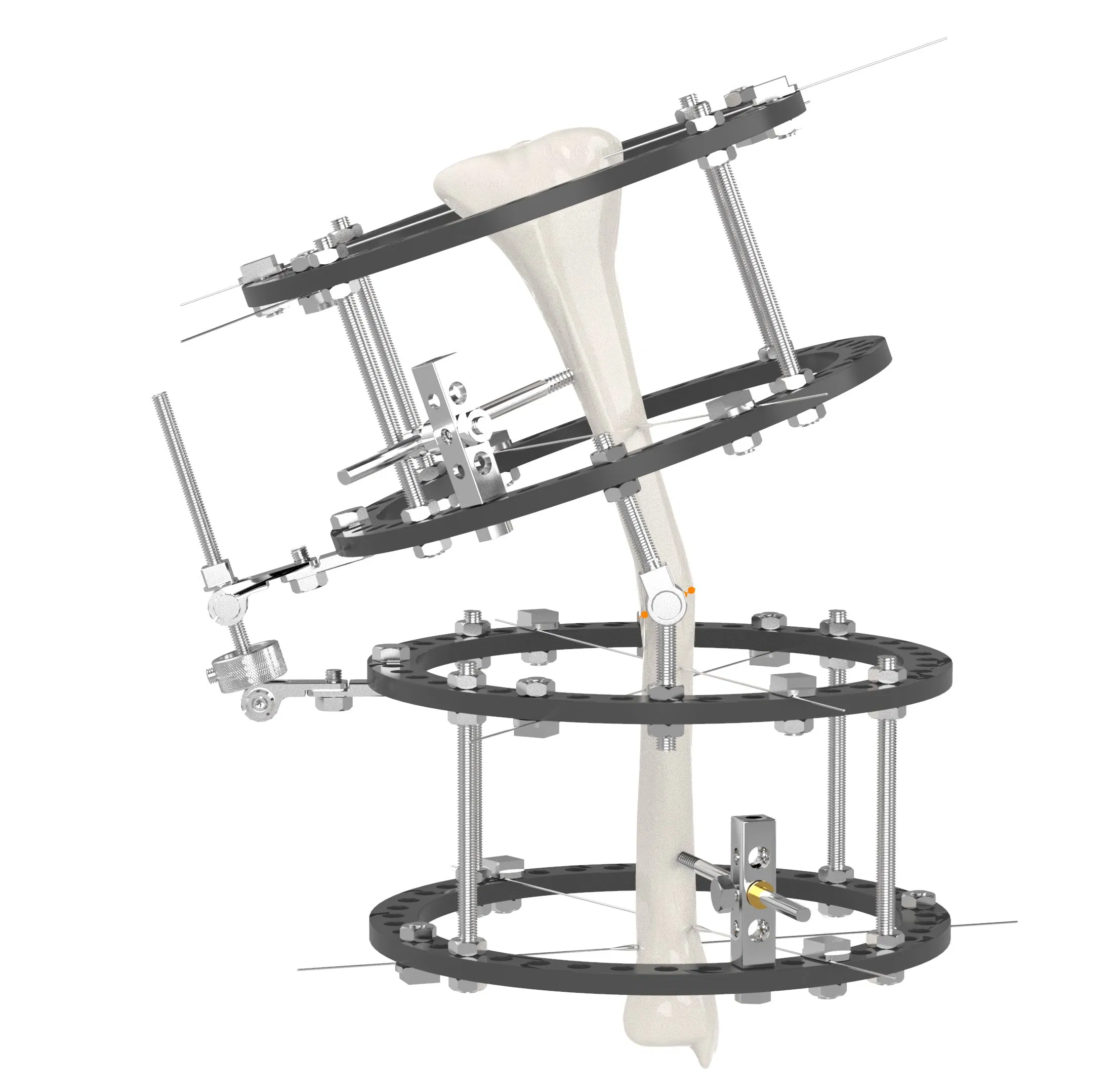
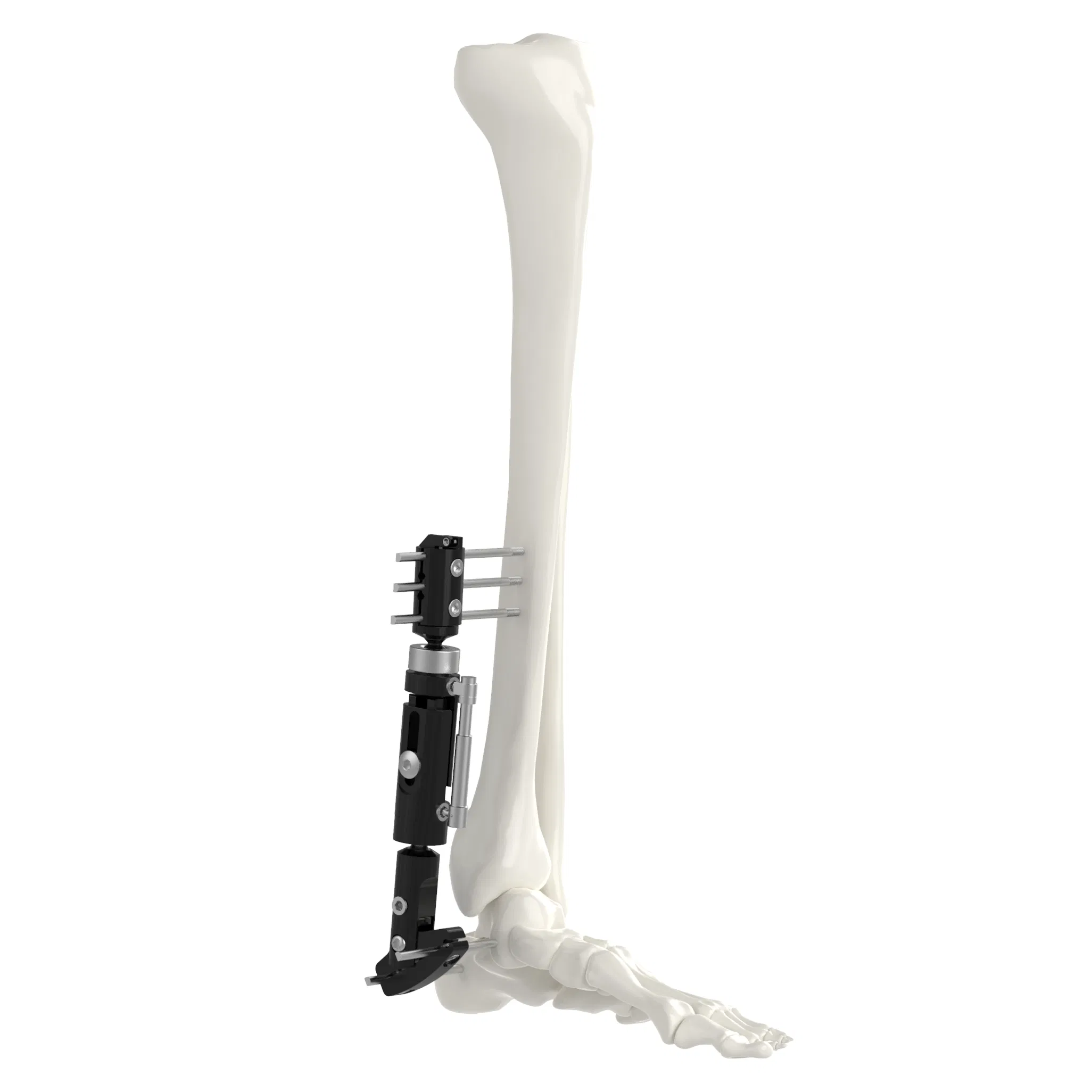
| System Components | |||
| Single arm | Combine | Ilizarov | Needle |




Founded in 1994, this medical device enterprise is currently one of the largest designers, manufacturers, and suppliers of orthopedic implants and instruments. The facility spans over 60,000 square meters of high-standard workshop space.
The core product lines encompass traumatology, spinal fixation systems, and specialized surgical instruments. Featured products include femoral nails, titanium plates, pedicle screws with rods, PEEK cages, and balloon kyphoplasty systems for spine fractures. These products are widely distributed across Europe, Latin America, the Middle East, and Asia.

 DTRX Orthopedic Implants
DTRX Orthopedic Implants