




1 / 5










| Still deciding? Get samples of $ ! US$ 7/Piece Request Sample |


Cannulated screws are a common method of fixation used by orthopedic surgeons for multiple fracture patterns. These screws are "hollow" to allow them to be placed over a guidewire facilitating better alignment prior to drilling or screw insertion. The guidewire is then removed and the screw is left in place. Options include headless and with-head types, available in sizes 3.0, 4.0, 6.5, and 7.3.

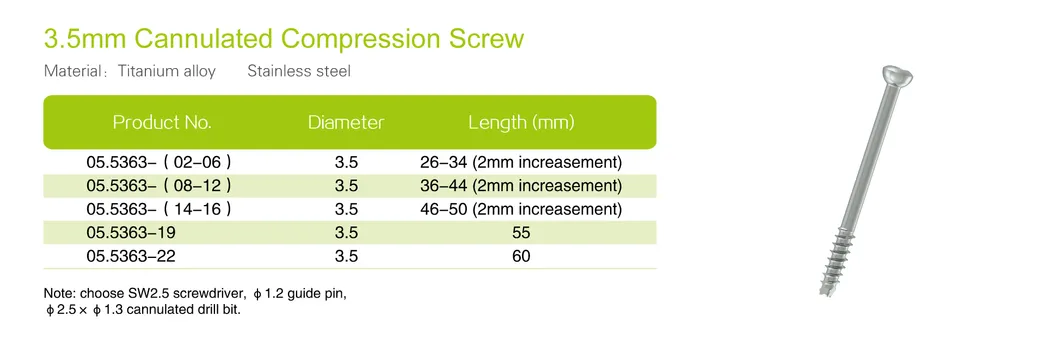


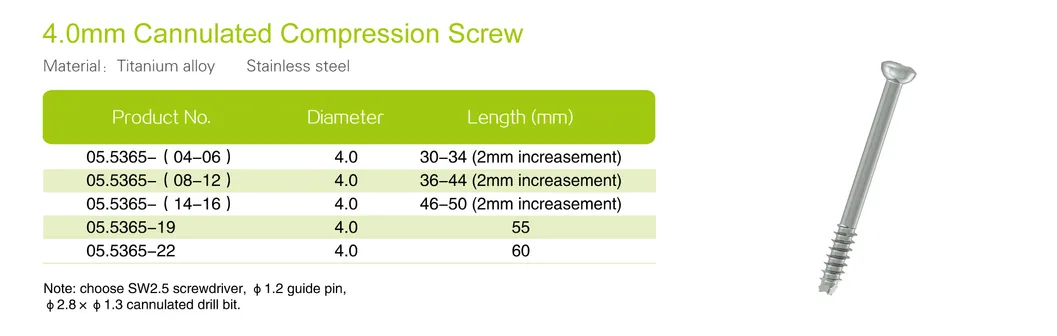

| Product No. | Diameter (mm) | Length (mm) |
|---|---|---|
| 05.5369-03 | 7.3 | 60 |
| 05.5369-04 | 7.3 | 65 |
| 05.5369-05 | 7.3 | 70 |
| 05.5369-06 | 7.3 | 75 |
| 05.5369-07 | 7.3 | 80 |
| 05.5369-08 | 7.3 | 85 |
| 05.5369-09 | 7.3 | 90 |
| 05.5369-10 | 7.3 | 95 |
| 05.5369-11 | 7.3 | 100 |
| 05.5369-12 | 7.3 | 105 |
| 05.5369-13 | 7.3 | 110 |
| 05.5369-14 | 7.3 | 115 |
| 05.5369-15 | 7.3 | 120 |




*All the prices are only for reference. Exact price will be based on the latest quotation.
 DTRX Orthopedic Implants
DTRX Orthopedic Implants