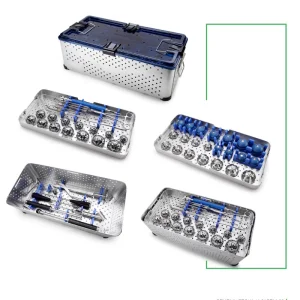

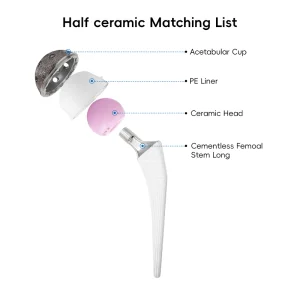

Leading the Global Market in High-Precision Revision Hip and Knee Arthroplasty
Switzerland stands at the epicenter of global medical technology, particularly in the domain of Revision Hip and Knee Systems. As a primary factory and exporter, our presence in the Swiss market is defined by a commitment to the highest industrial standards. Switzerland’s MedTech sector is world-renowned for its integration of micro-mechanics, material science, and digital health. In the context of joint revision surgery—a procedure required when a primary implant fails due to aseptic loosening, infection, or wear—the precision of the components is paramount.
Swiss manufacturing is synonymous with micron-level accuracy. Our revision systems leverage this heritage to ensure that bone-to-implant interfaces are optimized for long-term stability in complex secondary surgeries.
Utilizing high-grade Titanium alloys and UHMWPE (Ultra-High Molecular Weight Polyethylene), our products are designed to mimic the biomechanical properties of natural bone, reducing the risk of stress shielding.
The industrial landscape in cities like Zurich, Geneva, and Basel has fostered a cluster of innovation where research universities collaborate directly with orthopedic manufacturers. As a revision hip/knee systems factory, we tap into this ecosystem to incorporate cutting-edge developments like 3D-printed porous titanium structures. These structures allow for superior biological fixation, which is critical when the patient's existing bone stock is compromised.
Currently, the trend in Switzerland and globally is shifting toward Personalized Revision Surgery. This involves using 3D imaging and AI-driven planning to create custom-fit augments and stems. Our export operations ensure that these high-tech solutions reach hospitals worldwide, from the specialized clinics in the Swiss Alps to major surgical centers in North America and Asia. The demand for revision systems is growing due to the aging global population and the fact that primary implants are being placed in younger, more active patients who will eventually require a second procedure.
Our Swiss-based production lines utilize AI algorithms to monitor quality control in real-time, ensuring that every femoral stem and acetabular cup meets the rigorous CE and ISO 13485 certifications. This ensures zero-defect exports to our international partners.
In local hospitals like the Balgrist University Hospital in Zurich, our revision systems are applied in complex cases involving massive bone loss. The modularity of our systems allows surgeons to adapt the implant to the unique anatomy of each patient during the operation.
The sustainability of medical devices is also a major topic in the Swiss industrial sector. As a responsible exporter, we focus on biocompatible materials that reduce the risk of inflammatory responses. The "Swiss Made" label in orthopedics is not just about origin; it is a promise of durability and reliability. Our revision hip systems, for instance, utilize advanced taper designs and locking mechanisms that have been tested under extreme physiological loads to simulate 20+ years of use.
Precision Instruments and Implants Tailored for Swiss Standards







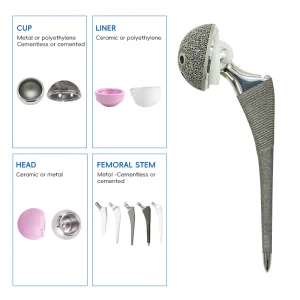
Partnering with Hangzhou DTRX Medical Technology for World-Class Orthopedics
Welcome to Hangzhou DTRX Medical Technology Co., Ltd. Our company is specialized in developing production and sales of orthopedic implants and instruments, serving as a vital node in the global supply chain, including the specialized Swiss MedTech market. Our factory covers a ground space of 32,068 square meters, equipped with a strong group of engineers and technicians dedicated to surgical excellence.
We satisfy the customers' requirements with consistent quality and favorable service, based on advanced production & processing technology and the best testing equipment imported directly from Germany. This ensures our products meet the "Switzerland standard" of precision and durability.

Employees
Plant Area
Volume
Patents
Our main product range includes: Spinal fixation systems, interlocking intramedullary nails, metal bone plates, metal bone screws, and related operation instruments. We take pride in doing business with customers from over 50 countries across the Middle East, Africa, South-east Asia, East Europe, Australia, North America, and South America. At the heart of our operations, we remain committed to the idea of "Care First." Innovation is our passion, and we look forward to bringing Swiss-level orthopedic precision to your medical facility. Believe in us, and you will be better!
One of the most significant hurdles in revision surgery is the management of bone loss. In Switzerland, surgical techniques often involve the use of impaction bone grafting or high-porosity metal augments. Our factory specializes in creating modular components that can bridge these defects. The modularity allows for the independent adjustment of leg length, offset, and version, which is vital for restoring the patient's biomechanics.
The Swiss market demands materials that minimize metallosis and periprosthetic osteolysis. To meet this, our revision systems undergo rigorous fatigue testing. We employ advanced surface treatments, such as plasma-sprayed hydroxyapatite (HA) coatings, to promote rapid osseointegration. This technology ensures that even in the compromised environment of a revision surgery, the implant can achieve a stable, long-term bond with the host bone.
As a leading exporter, we understand that different regions have varying regulatory and clinical requirements. However, the "Swiss Quality" benchmark serves as a universal language of trust. Whether it is a hospital in Geneva or a clinic in New York, the expectation for a revision hip or knee system is the same: it must be intuitive for the surgeon and life-changing for the patient. Our instrument sets are designed with ergonomic handles and simplified tray layouts to reduce "the footprint" in the OR and decrease surgery time.
Looking ahead, the integration of robotic-assisted surgery is the next frontier. Switzerland is currently home to some of the most advanced research in surgical robotics. Our revision systems are being developed to be "robot-ready," meaning they are compatible with navigation systems that provide real-time feedback to the surgeon. This ensures that the placement of a revision stem or cup is accurate to within a millimeter, significantly reducing the risk of future failures.
Choosing a Revision Hip/Knee Systems factory and exporter means choosing a partner in patient care. Our commitment to Swiss-quality engineering, combined with our large-scale manufacturing capabilities in Hangzhou, allows us to provide high-end solutions at a competitive scale. We continue to innovate, ensuring that every patient who receives one of our implants can return to an active, pain-free life.