Advanced Craniomaxillofacial (CMF) Solutions & Trauma Fixation Systems Tailored for West African Healthcare Excellence
Establishing a new paradigm in surgical precision and titanium implant durability.
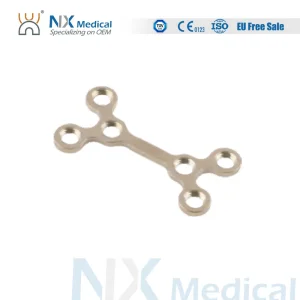
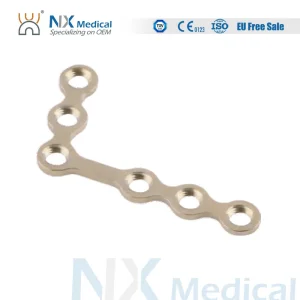
In Guinea-Bissau, the demand for maxillofacial surgery is rising significantly. Rapid urbanization and transport challenges have led to an increase in craniofacial trauma cases. Hospitals in Bissau are increasingly seeking high-quality, ISO 13485 certified titanium implants that offer both biological compatibility and mechanical stability. As a leading manufacturer, we provide the specific anatomical plates required for the complex reconstructive needs of West African patients.
International medical procurement for the African market has shifted towards "Value-Based Healthcare." Buyers are no longer looking for the cheapest option but for Information Gain—products that provide superior clinical outcomes, such as our Variable Angle (VA) LCP systems. Our export division ensures seamless logistics to the Port of Bissau, providing comprehensive documentation for medical device clearance.
With over 20 years of experience, Hangzhou DTRX Medical Technology (Youbetter) demonstrates unparalleled Expertise and Authority. Our facility, covering 32,068 sqm, utilizes German-imported testing equipment to ensure every maxillofacial screw and plate meets the rigorous CE certification standards, ensuring Trustworthiness for every surgeon in Guinea-Bissau.
Hangzhou DTRX Medical Technology Co., Ltd. is specialized in developing production and sales of orthopedic implants and instruments. Our factory satisfies the customers' requirements with satisfied quality and favorable service, based on advanced processing technology.
Transitioning from standard Titanium Alloy to PEEK (Polyether ether ketone) for specific trauma cases where radiolucency is critical. This is vital for Guinea-Bissau's growing oncology and reconstruction clinics.
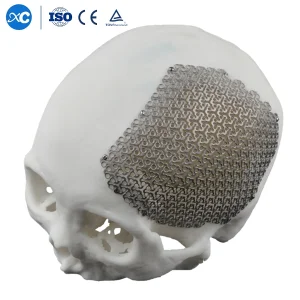
Utilizing 3D cloud-shape anatomical mesh for cranial defects. Our 3D printing capabilities allow for patient-specific implants that reduce surgery time and improve aesthetic outcomes.
Our VA-LCP systems allow screws to be angled up to 15°, providing surgeons in Bissau the flexibility to avoid vital structures like tooth roots or nerves while maintaining rigid fixation.

Precision Machining

Turning And Milling Process

Finish Machining

Five Axis Grinding

CNC Machining Center

Hardness Testing Machine
As a global exporter, we understand the regulatory landscape of Guinea-Bissau. We provide:
Regulatory Support
Full CE & ISO Documentation
Sterile Packaging
Double-layer sterile packing
Rapid Logistics
Express air & sea freight
Surgical Training
Remote and on-site support
We primarily use high-grade Titanium Alloy (Ti-6Al-4V) and PEEK. These materials are chosen for their excellent biocompatibility, strength-to-weight ratio, and long-term stability in the human body.
Yes, we specialize in 3D anatomical cloud-shape cranial mesh and customized plates based on CT scan data, allowing for precise reconstruction of complex facial defects.
Standard air freight usually takes 7-10 days, while sea freight via the Port of Bissau takes approximately 35-45 days depending on the shipping line. All products are shipped with full export documentation.
Absolutely. Hangzhou DTRX Medical is ISO 9001 and ISO 13485 certified. Our products have passed CE certification, ensuring they meet the highest international safety standards.