Leading surgical implants designed for Panama's top orthopedic clinics.
Panama has rapidly emerged as a strategic cornerstone for medical device distribution and advanced surgical procedures in Central and South America. As the "Hub of the Americas," the country's unique geographical position, bolstered by the Panama Canal and the Tocumen International Airport, makes it an ideal center for the logistics of high-precision medical instruments like Intramedullary Nails.
The demand for orthopedic implants in Panama is driven by a sophisticated healthcare system that includes both the public Caja de Seguro Social (CSS) and a booming private sector centered around Panama City. Facilities like Punta Pacifica and Hospital Paitilla are increasingly seeking international-standard implants that provide faster recovery times for patients suffering from long-bone fractures.
Our factory utilizes German-imported testing equipment to ensure every Intramedullary Nail meets the rigorous mechanical standards required for Panama's diverse patient demographic.
Given Panama's humid climate, our Grade 5 Titanium alloys offer superior corrosion resistance and biocompatibility compared to traditional stainless steel alternatives.
Leveraging the Colón Free Trade Zone, we ensure that our orthopedic trauma systems reach Panamanian hospitals with minimal lead time and maximum supply chain transparency.
Welcome to Hangzhou DTRX Medical Technology Co., Ltd. Our company is specialized in developing production and sales of orthopedic implants and instruments. The factory covers a ground space of 32,068 square meters. Meanwhile we have a strong group of engineers and technicians. The company satisfies the customers' requirements with satisfied quality and favorable service, which is based on the advanced production & processing technology and the best testing equipment imported from Germany.
Core Advantages:

The orthopedic industry in Panama is witnessing a shift towards minimally invasive surgery (MIS). Intramedullary nailing has become the gold standard for femoral and tibial shaft fractures due to its biological advantages, such as preserving the periosteal blood supply and allowing early weight-bearing. In Panama City, where traffic-related trauma and industrial accidents are common, the need for reliable, interlocking nail systems is at an all-time high.
Modern hospitals in Panama are beginning to integrate robotic assistance and 3D imaging into the operating room. Our Expert Intramedullary Nails are designed to be compatible with advanced fluoroscopy and navigation systems, ensuring that Panamanian surgeons can achieve precise proximal and distal locking with minimal radiation exposure.
In the Panamanian market, there is a distinct preference for Titanium Alloy (Ti6Al4V). This is not only due to its lightweight nature but also its modulus of elasticity, which is closer to human bone compared to stainless steel. This reduces the "stress shielding" effect, promoting faster callous formation in patients treated at institutions like the Instituto de Ciencias Médicas.
Like many developing nations, Panama is seeing an increase in geriatric orthopedic cases. The PFNA (Proximal Femoral Nail Antirotation) is critical for treating intertrochanteric fractures in elderly Panamanian patients with osteoporotic bone. Our helical blade technology provides enhanced stability, which is vital for preventing secondary fractures and ensuring the patient returns to mobility quickly.
Explore our full range of CE-certified orthopedic solutions available for Panama.
 High-Quality Interlocking Nail Intramedullary Nail in Blue Green Gold Colors
High-Quality Interlocking Nail Intramedullary Nail in Blue Green Gold Colors
 Factory Instrument Expert Femur Intramedullary Nail Orthopedic Intramedullary Nails for Human
Factory Instrument Expert Femur Intramedullary Nail Orthopedic Intramedullary Nails for Human
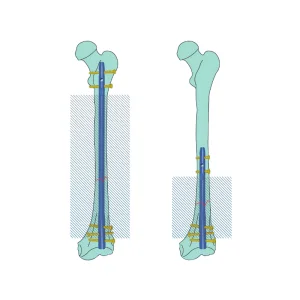 Czmeditech Orthopedic Expert Reversed Femoral Intramedullary Nail
Czmeditech Orthopedic Expert Reversed Femoral Intramedullary Nail
 Orthopedic Fixation with Retrograde Intramedullary Femur Interlocking Intramedullary Nails 11*200
Orthopedic Fixation with Retrograde Intramedullary Femur Interlocking Intramedullary Nails 11*200
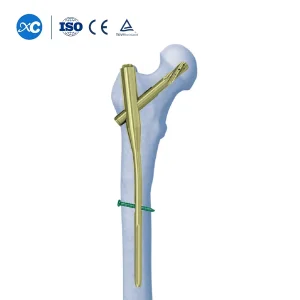 Price Gama Nails Pfna Surgery Nail Antirotation Titanium Femur Intramedullary Nail
Price Gama Nails Pfna Surgery Nail Antirotation Titanium Femur Intramedullary Nail
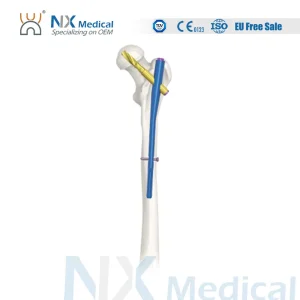 Nx Medical Orthopedic Implant Pfna Proximal Femoral Titanium Intramedullary Nail Bone Fixation
Nx Medical Orthopedic Implant Pfna Proximal Femoral Titanium Intramedullary Nail Bone Fixation
 Best Price Intramedullary Nail System Expert Tibial Proximal Interlocking Nail
Best Price Intramedullary Nail System Expert Tibial Proximal Interlocking Nail
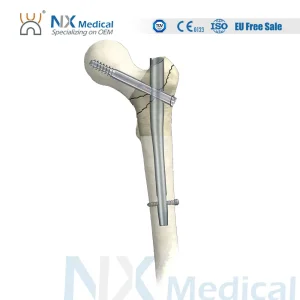 Nx Medical Orthopedic Trauma Titanium Intramedullary Interlocking Femoral Femur Pfna Bone Nail
Nx Medical Orthopedic Trauma Titanium Intramedullary Interlocking Femoral Femur Pfna Bone Nail
As a global manufacturer, we understand that providing Intramedullary Nails to Panama involves more than just shipping a product. It involves a commitment to education, quality assurance, and long-term partnership. Our products, including the Spinal fixation system, metal bone plates, and related operation instruments, have passed ISO 9001, ISO 13485, and CE certification.
We do business with customers from over 50 countries, and we are looking to expand our footprint in Panama by offering localized support and customized implant designs. Our R&D team continuously innovates to meet the specific anatomical needs of the Latin American population.
Believe in us, You will be better!