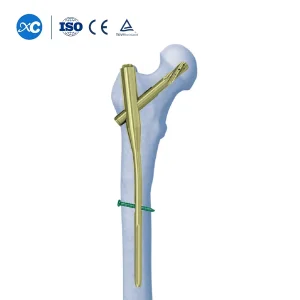
Price Gama Nails Pfna Surgery Nail Antirotation Titanium Femur Intramedullary Nail
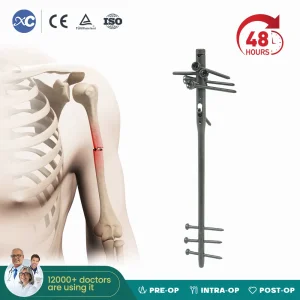

Canwell Tibial Nail Intramedullary Nail Interlocking Cannualted Nail Canetn Gamma Nail


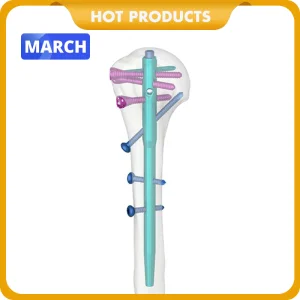

China Factory Nx Medical Orthopedic Trauma Implant Titanium Retrograde Tibial Intramedullary Nail
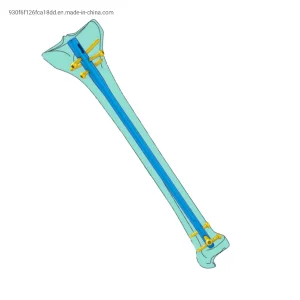
Intramedullary Nail System Expert Tibial Interlocking Nail Suprapatellar Approach
The global orthopedic trauma market is witnessing a significant paradigm shift, with the Tibial Intramedullary Nail market emerging as a cornerstone of modern fracture management. As the gold standard for treating tibial shaft fractures, these implants are integral to the $50 billion global orthopedic industry. Currently, the market is driven by an aging population prone to osteoporosis-related fractures and a rise in high-energy trauma incidents such as road traffic accidents and sports injuries.
Industrial production of these medical devices has evolved from simple stainless steel rods to high-precision Titanium Alloy (Ti-6Al-4V ELI) systems. As a leading OEM/ODM Tibial Intramedullary Nail factory, we recognize that the global demand is no longer just for basic implants, but for sophisticated systems that offer biological fixation, multi-planar locking options, and anatomical pre-bending. The market is currently dominated by North America and Europe, but the fastest growth is seen in the Asia-Pacific and Latin American regions, where healthcare infrastructure is expanding rapidly.
The transition from infrapatellar to suprapatellar approach is a major trend. It allows for easier reduction of proximal fractures and reduces anterior knee pain, a common post-operative complication.
Technological trends are leaning towards "Smart Nails" and robotic-assisted distal locking. Our R&D department is focusing on instrumentation compatible with navigation systems to reduce radiation exposure.
Developing bioactive coatings to accelerate bone healing and reduce infection rates (Anti-bacterial coatings) is the next frontier for orthopedic implant exporters.
Global hospital groups and medical distributors face the challenge of balancing high-quality surgical outcomes with cost-containment. Our Tibial Intramedullary Nail systems are designed to meet diverse localized needs:
Localized application is particularly critical in regions like Southeast Asia and the Middle East, where high-speed transport infrastructure has increased the incidence of complex open fractures requiring immediate intramedullary stabilization.

As a prominent OEM/ODM Tibial Intramedullary Nail Exporter based in Hangzhou, China, we offer a unique blend of technological innovation and manufacturing efficiency. Our competitive edge is built on four pillars:
Utilizing German-imported testing equipment and high-precision CNC machining centers to ensure every nail meets micron-level tolerances.
Our facility is ISO 9001, ISO 13485, and CE certified, ensuring seamless entry into over 50 international markets.
We provide full-spectrum ODM services, from initial 3D design and prototyping to private labeling and custom packaging.
Welcome to Hangzhou DTRX Medical Technology Co., Ltd. Our company is specialized in developing production and sales of orthopedic implants and instruments. The factory covers a ground space of 32,068 square meters. Meanwhile we have a strong group of engineers and technicians. The company satisfies the customers' requirements with satisfied quality and favorable service, which is based on the advanced production & processing technology and the best testing equipment imported from Germany.
We Provide Exceptional Solutions for Clients:
Enterprise has passed ISO 9001 and ISO 13485 system certification, products have passed the CE certification. We do business with customers from over 50 countries from Middle east, Africa, South-east Asia, East Europe, Australia, North America, South America. Youbetter remains the ideas of "care first", and we pay much attention to developing the new products, improving the quality of the products, and hankering the perfect of the service. We have a strong team of researching and developing new products which can meet the different needs of different customers. We keep innovation and have full of passion to be better. We are looking forward to having business with you in near future! Believe in us, You will be better!
In the realm of tibial intramedullary nail manufacturing, material selection and mechanical design are paramount. Our products utilize Grade 5 Titanium (Ti-6Al-4V), offering superior fatigue strength and modulus of elasticity closer to cortical bone compared to stainless steel. This reduces the "stress shielding" effect, promoting faster callous formation.
Our design incorporates a 10° proximal bend to facilitate insertion and a distal locking configuration that allows for static or dynamic compression. The cannulated design supports reamed or unreamed techniques, providing surgeons with maximum intraoperative flexibility. As a dedicated exporter of orthopedic surgical equipment, we ensure that our instrumentation sets are ergonomically designed, reducing "surgeon fatigue" and operative time.

Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail

Expert Tibial Intramedullary Orthopedic Titanium Tibia Interlocking Nail
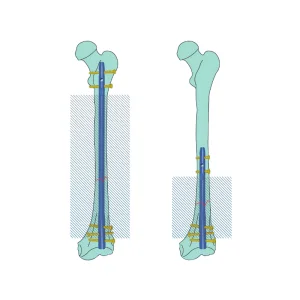
Czmeditech Orthopedic Expert Reversed Femoral Intramedullary Nail

Best Price Intramedullary Nail System Expert Tibial Proximal Interlocking Nail

High-Quality Interlocking Nail Intramedullary Nail in Blue Green Gold Colors
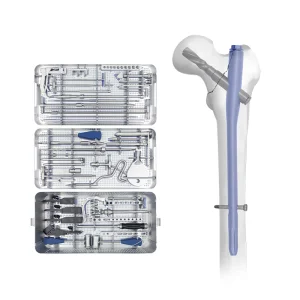
Medical Supply Orthopedic Implant Titanium Femur Pfna Intramedullary Nail Instrument Set


Intramedullary Nail System Expert Humeral Proximal Interlocking Nail Blade Locking