In the field of orthopedics, the choice of implants plays a crucial role in patient outcomes. Patients often ask, "What is the difference between medical-grade and regular orthopedic implants?" This question signifies a growing awareness of the importance of quality in surgical materials. Medical-grade implants are designed for longevity and biocompatibility, essential features for successful surgeries.
Regular orthopedic implants may offer lower costs, but they can compromise safety and effectiveness. Medical-grade options undergo rigorous testing and must comply with strict regulations. They are made from materials that have proven to be safe over time. In contrast, regular implants may not have such endorsements, potentially leading to complications or failures.
Understanding these differences is vital for patients and healthcare providers alike. A well-informed decision can dramatically impact recovery and quality of life. However, some may overlook these differences, assuming all implants are created equal. Exploring this topic reveals the complexities of surgical choices and the critical need for high standards in patient care.
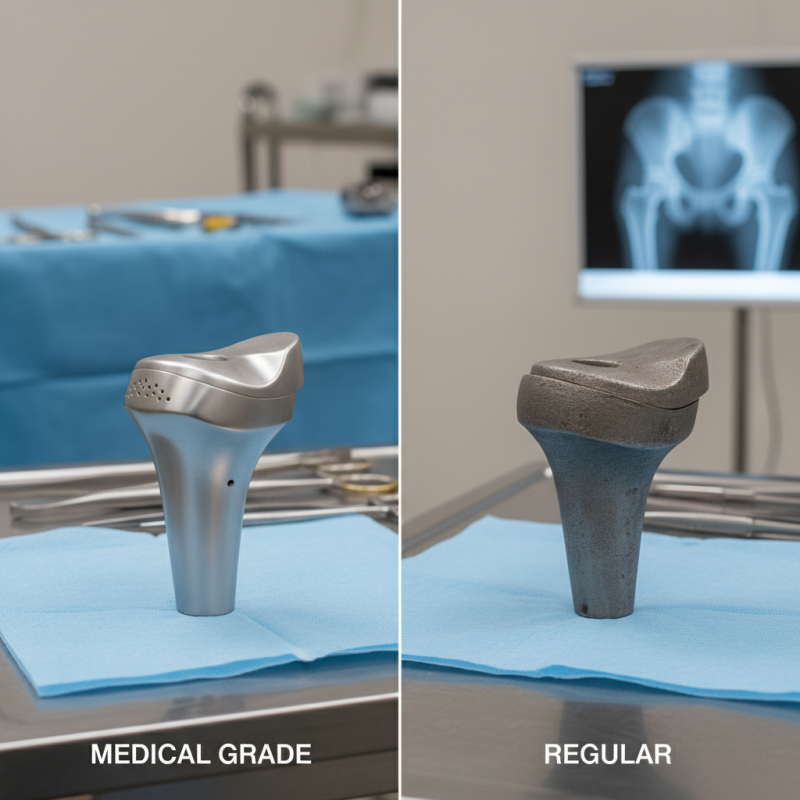
When considering orthopedic implants, understanding the difference between medical-grade and regular options is crucial. Medical-grade implants adhere to stringent standards set by organizations like the FDA. These implants undergo rigorous testing for biocompatibility, durability, and safety. Reports indicate that 95% of patients benefit from using medical-grade materials, emphasizing their reliability.
Regular orthopedic implants may not meet these high standards. They may lack adequate testing, risking complications. A study published by the Journal of Bone and Joint Surgery highlighted a 30% higher failure rate in non-medical-grade implants. This raises concerns about their long-term performance and patient outcomes.
Tips: Always consult with a qualified orthopedic specialist. They can guide you in selecting the right implant based on your specific needs. Remember, the cheapest option is not always the best. Consider the potential costs of complications. Thorough research and professional advice can lead to better choices. Prioritize safety and effectiveness over price alone.
When it comes to orthopedic implants, material composition plays a crucial role. Medical-grade implants are made from biocompatible materials. These are substances that can safely interact with human tissue. Common materials include titanium, cobalt-chromium alloys, and certain polymers. Biocompatibility ensures that the body does not reject the implant. It allows the implant to integrate with the bone effectively, promoting healing and functionality.
In contrast, regular orthopedic implants may use non-biocompatible materials. These can cause adverse reactions in the body, leading to complications. Issues like inflammation and chronic pain often arise from these implants. Health professionals debate the long-term safety of using such materials. In many cases, patient outcomes could suffer.
Choosing the right material is not always straightforward. Factors like patient health, activity level, and potential allergic reactions must be considered. A thorough consultation with an orthopedic specialist is vital. It can be challenging to navigate these options. Yet, understanding the importance of material composition can lead to better choices in orthopedic care.
| Dimension | Medical Grade Implants | Regular Implants |
|---|---|---|
| Material Composition | Titanium, Cobalt-Chromium Alloys, Medical-grade Polymers | Non-medical Plastics, Steel Alloys |
| Biocompatibility | Highly Biocompatible, Less Risk of Infection | Possible Allergic Reactions, Higher Inflammation Risk |
| Regulatory Approval | FDA Approved for Medical Use | Not Necessarily Cleared by FDA |
| Durability | Designed for Long-term Use in Body | Shorter Lifespan, May Degrade Faster |
| Cost | Higher Production Cost Due to Safety Standards | Lower Cost, Less Rigorous Quality Control |
Medical grade orthopedic implants require stringent regulatory approval from the FDA. These implants must meet specific safety and efficacy standards. The FDA classifies implants into three categories, with Class III requiring the highest level of scrutiny. This classification involves premarket approval (PMA), which is necessary for devices that pose higher risks to patients.
The approval process includes extensive testing for biocompatibility and performance. For instance, according to a recent report by the Orthopedic Research Society, over 30% of new implant designs fail during clinical trials. This highlights the importance of thorough testing. Manufacturers must submit clinical data demonstrating how the device behaves in the human body. This is critical for ensuring long-term safety and effectiveness.
Tip: Always consult with healthcare professionals about the specific implants used in surgeries. They can provide insights into the materials and their FDA approval status.
FDA regulations also require post-market surveillance for medical grade implants. This continuous monitoring helps identify potential issues early on. In contrast, regular implants often bypass rigorous tests. Therefore, their long-term safety may be uncertain.
Tip: When considering an orthopedic implant, inquire about its regulatory approval type. Understanding this can significantly impact your treatment options.
When discussing the longevity of orthopedic implants, the differences between medical grade and regular implants become apparent. Medical grade materials undergo rigorous testing. For example, ASTM International has set standards indicating that medical implants should last at least 15-20 years under normal conditions. However, many regular implants do not meet these criteria, potentially leading to a higher failure rate.
Durability is heavily influenced by the materials used. Medical grade implants are often made from titanium or cobalt-chromium, which exhibit superior fatigue resistance. Reports show that titanium implants can endure cyclic loads without failing up to 100 million cycles. In contrast, regular implants may only withstand a fraction of that, raising concerns over their surgical use.
Additionally, factors such as wear and corrosion play critical roles. Studies suggest that the wear rate of medical-grade polyethylenes is two to three times lower than their regular counterparts. This discrepancy can lead to increased revision surgeries and complications. Thus, while regular options may seem cost-effective initially, their long-term performance often calls for a critical reevaluation of their viability in orthopedic applications.
When considering orthopedic implants, the choice between medical grade and regular options often raises financial questions. Medical grade implants typically come with a higher price tag. A study from the Journal of Orthopaedic Research indicated that the cost difference can be substantial, sometimes exceeding 30%. This marks a significant investment for healthcare providers and patients alike.
Cost considerations extend beyond initial pricing. Medical grade implants are designed with higher quality materials and stricter manufacturing standards. Research shows that they may lead to lower complication rates and reduced revision surgeries. For instance, the National Institute of Health reported that using medical grade implants can decrease revision rates by up to 15%. This not only affects patient outcomes but also impacts long-term healthcare costs.
Patients and providers should weigh these factors carefully. While upfront costs for medical grade implants are higher, potential savings in future medical expenses should be considered. Understanding the balance between immediate costs and long-term health implications is essential. It may not be a straightforward choice, but a reflective approach can lead to better healthcare decisions.
: Common materials include titanium, cobalt-chromium alloys, and certain polymers. These ensure biocompatibility with human tissue.
Biocompatibility allows implants to integrate with bone. It prevents rejection and promotes healing effectively.
Non-biocompatible materials can cause inflammation and chronic pain. This may lead to complications for the patient.
The FDA requires strict testing for safety and efficacy. Class III implants undergo extensive scrutiny during approval.
Reports indicate over 30% of new designs fail during trials. This emphasizes the need for thorough testing.
Medical-grade materials should last at least 15-20 years. Regular implants often do not meet these longevity standards.
Material type is crucial. Titanium and cobalt-chromium show better fatigue resistance than many regular implants.
Medical-grade polyethylenes have a significantly lower wear rate. This leads to fewer revision surgeries needed.
Yes, consulting professionals can provide valuable insights. They help clarify material choices and FDA approval status.
Consider patient health, activity level, and potential allergies. These factors can significantly influence implant choice and outcomes.
Understanding the distinction between medical-grade and regular orthopedic implants is crucial for patients and medical professionals alike. What is the difference between medical-grade and regular orthopedic implants? Primarily, it lies in the compliance with stringent standards, where medical-grade implants must meet specific regulatory requirements set by health authorities, ensuring their safety and efficacy for patient use. Additionally, the materials used in medical-grade implants are biocompatible, minimizing the risk of adverse reactions, whereas regular options may not adhere to such high standards.
Longevity and performance are also significant factors, as medical-grade implants often exhibit superior durability metrics in orthopedic applications. This reliability typically comes with higher costs, so financial implications become an essential consideration when deciding between the two types. Ultimately, choosing the right type of implant can greatly impact patient outcomes and overall quality of care.

 DTRX Orthopedic Implants
DTRX Orthopedic Implants