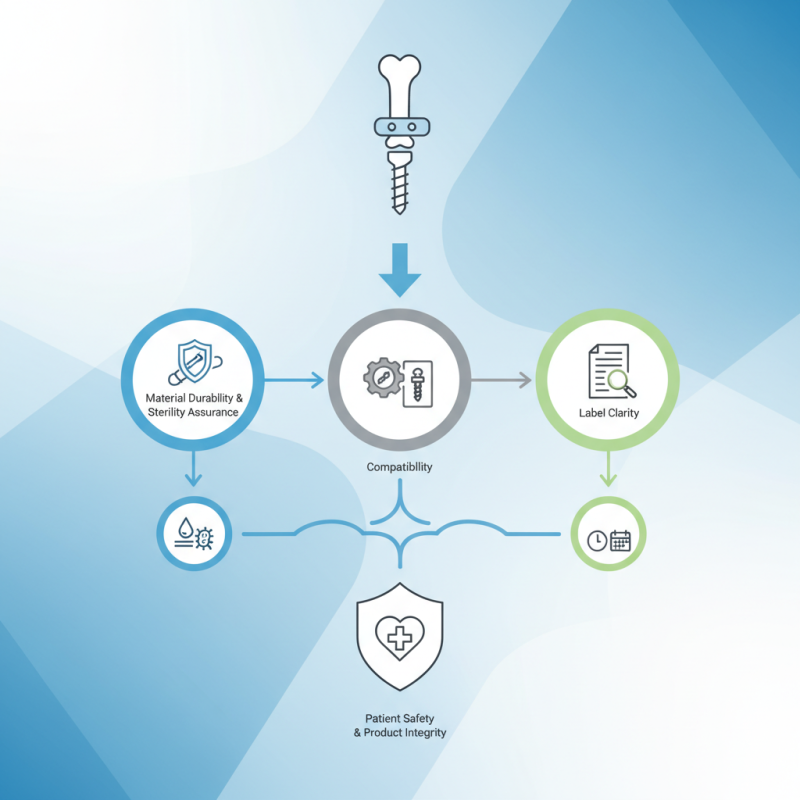
Choosing the right orthopedic implants packaging is crucial for patient safety and product integrity. The packaging safeguards delicate implants from contamination and damage. Understanding what to look for in orthopedic implants packaging can streamline your selection process.
Key factors include material durability and sterility assurance. Packaging should withstand transportation and storage while maintaining a sterile environment. It is essential to consider the compatibility between the packaging materials and the implants themselves. Improper selection can lead to compromised implants and affect surgical outcomes.
Additionally, label clarity is vital. Clear instructions and expiration dates help healthcare professionals ensure proper storage and usage. However, not all packaging meets these requirements, which can lead to costly mistakes. Evaluating different packaging options critically is necessary. The implications extend beyond the operating room to patient health. Therefore, making informed decisions about orthopedic implants packaging is imperative.
Choosing the right packaging for orthopedic implants requires careful consideration of several factors.
The packaging must maintain sterility, ensuring that the implants remain free of contamination.
A report by the FDA highlights that improper packaging can lead to increased infection rates, underscoring the need for
robust materials and processes.
Durability is critical. The packaging should protect implants from physical damage during transport.
According to industry studies, packaging failures account for up to 20% of product recalls.
Additionally, traceability is essential. Packaging must include proper labeling to ensure compliance with regulatory standards.
Clear identification helps in tracking and managing the implants throughout their lifecycle.
The environmental impact also cannot be ignored. The industry is moving towards sustainable packaging solutions.
Using recyclable materials not only helps the planet but can also appeal to hospitals seeking greener options.
However, transitioning to eco-friendly options can be challenging. Balancing sustainability with safety and efficacy requires
innovative solutions and ongoing reevaluation of materials.
When selecting the right packaging for orthopedic implants, materials play a crucial role. Various packaging materials are used to ensure safety and integrity. Commonly, high-density polyethylene (HDPE) is favored for its durability and resistance to moisture. This makes it a reliable option for sterile environments. Polyvinyl chloride (PVC) is another popular choice, known for its flexibility and transparency. It allows for easy visual inspection of the product inside.
Beyond plastics, other materials are worth considering. Paper and cardboard packaging can provide a sustainable alternative. They are often used in combination with coatings to enhance moisture resistance. However, reflective surfaces can sometimes complicate inspections. Balancing visibility and protection is key. Materials also need to withstand handling during shipping and storage.
It’s critical to evaluate the compatibility of the packing material with the implants. Some materials might interact negatively with specific implants. This potential risk requires careful assessment. Manufacturers often test different materials to ensure that they do not compromise the integrity of the implants. The right packaging material not only protects but also supports the functionality of the orthopedic implants throughout their lifecycle.
| Packaging Material | Benefits | Common Uses | Considerations |
|---|---|---|---|
| Blister Packaging | Provides visibility and protects against contamination. | Screws, plates, and small implants. | Must ensure proper sealing to maintain sterility. |
| Rigid Containers | Durable and reusable, offering excellent protection. | Instruments and larger implants. | Heavier and may be more expensive to transport. |
| Foam Inserts | Customizable protection and cushioning for fragile items. | Intricate and delicate implants. | Need to ensure compatibility with sterilization methods. |
| Pouch Packaging | Lightweight and space-efficient. | Surgical kits and individual instruments. | May not provide as much protection as rigid options. |
| Sealed Trays | Ensures sterility and minimizes handling. | Complete surgical sets. | More complex to manufacture and may increase costs. |
The importance of sterility cannot be overstated in the packaging of orthopedic implants. These implants come in direct contact with the human body. Any contamination could lead to serious infections. Packaging must ensure that these devices remain sterile until the moment they're needed.
Designing for safety is crucial. Materials used should be durable yet allow for sterilization. Manufacturers often utilize barrier technologies to protect against moisture and air. However, not all packaging designs achieve this effectively. It is vital to assess how well the packaging withstands environmental stressors.
Reflecting on current practices, there's a need for improvement. Some packages may not offer adequate protection during transportation. This can compromise sterility. Continuous testing and quality assurance protocols are essential. Regular reviews help identify weaknesses in current designs, paving the way for innovation and enhanced safety measures.
When it comes to orthopedic implants packaging, regulatory standards play a crucial role. These standards ensure patient safety and product integrity. Various organizations set regulations that must be adhered to during the packaging process. These guidelines cover materials, sterility, labeling, and more.
Manufacturers must understand and implement these standards thoroughly. Failure to comply can result in severe consequences. For instance, improper sterilization can lead to infections. Inadequate labeling might confuse healthcare providers. Both scenarios compromise patient care. There's a continuous need to evaluate and improve compliance procedures.
Moreover, keeping up with the evolving regulations is challenging. New insights in materials science may reshape standards. Companies must stay informed about these changes. This involves attending conferences and engaging with regulatory bodies. Sometimes, errors in the packaging process also occur, pushing a reevaluation of practices. These reflections are necessary for growth and advancement in the industry.
This chart represents the importance level of various factors to consider when choosing the right packaging for orthopedic implants, based on regulatory standards. Material quality and regulatory compliance are the top priorities.
Sustainability is a pressing concern in many industries, including orthopedic implants. The packaging of these medical devices plays a crucial role in reducing environmental impact. A recent report from the Medical Device Innovation Consortium emphasizes that sustainable packaging can decrease carbon footprints by over 30%, highlighting the importance of materials selection.
Life cycle assessments show that packaging materials significantly affect product sustainability. For instance, switching to bioplastics can potentially cut down plastic waste. However, many manufacturers hesitate due to cost concerns and performance standards. The challenge lies in balancing eco-friendly options with compliance regulations.
There’s a growing recognition of the need to rethink traditional packaging methods. Minimalism in design and packaging can lead to reductions in waste. Yet, the industry sometimes prioritizes convenience over sustainability. Stakeholders must engage and discuss best practices to ensure that sustainability does not compromise safety and efficacy.
: High-density polyethylene (HDPE) and polyvinyl chloride (PVC) are commonly used materials.
Durability ensures that packaging can withstand moisture and handling during shipping.
Transparency allows for easy product inspection but may complicate moisture resistance.
Yes, they can be sustainable alternatives, but may require coatings for better moisture resistance.
Sterility is crucial, as any contamination can lead to serious infections.
By using durable materials and barrier technologies to protect against air and moisture.
Some designs may not provide adequate protection during transportation, risking sterility.
It helps identify weaknesses in packaging and supports innovation in safety measures.
Yes, compatibility must be assessed to avoid potential negative interactions.
Balancing visibility and protection while ensuring materials are eco-friendly.
Choosing the right orthopedic implants packaging is crucial for ensuring the safety and effectiveness of the implants. When considering what to look for in orthopedic implants packaging, several key factors come into play. First, it is essential to evaluate the types of packaging materials used, ensuring they provide adequate protection and maintain sterility. Additionally, the design must prioritize safety, aligning with regulatory standards to guarantee compliance and minimize risk to patients.
Sustainability is also an important consideration in the packaging choices for orthopedic implants, as healthcare increasingly focuses on eco-friendly practices. By selecting materials and designs that reduce environmental impact, manufacturers can enhance the overall sustainability of their products. Ultimately, thoughtful consideration of these factors will result in packaging that not only protects the implants but also supports broader health and environmental goals.

 DTRX Orthopedic Implants
DTRX Orthopedic Implants