A trusted global manufacturer with over 20 years of expertise in orthopedic implants — delivering precision-engineered solutions to surgeons and distributors across 50+ countries.
Welcome to Hangzhou DTRX Medical Technology Co., Ltd. Our company is specialized in developing, producing, and selling orthopedic implants and instruments. The factory covers a ground space of 32,068 square meters. We have a strong group of engineers and technicians who satisfy customers' requirements with superior quality and dedicated service — backed by advanced production & processing technology and the best testing equipment imported from Germany.

Our main products are engineered to meet the demands of modern orthopedic surgery — from complex spinal reconstructions to trauma fixation. Every implant is manufactured under strict ISO 13485 quality management protocols.
Comprehensive pedicle screw systems, rods, and connectors designed for thoracic, lumbar, and cervical spine stabilization. Available in titanium alloy (Ti-6Al-4V) and stainless steel 316L.
Precision-engineered femoral, tibial, and humeral nails with proximal and distal locking options. Designed for minimal soft tissue disruption and rapid patient recovery.
Dynamic compression plates (DCP), locking compression plates (LCP), and reconstruction plates for long bone and periarticular fractures. Low-profile designs reduce soft tissue irritation.
Cortical, cancellous, cannulated, and locking screws in a full range of diameters and lengths. Self-tapping thread geometry ensures secure fixation in diverse bone densities.
Complete sets of related operation instruments including drill guides, depth gauges, targeting arms, and torque-limiting screwdrivers — all reusable and autoclave-compatible.
Patient-specific and OEM implants designed to your drawings or specifications. Our R&D team collaborates closely with distributors and hospitals to bring custom solutions to market.
From raw material traceability to post-delivery technical support, DTRX is committed to being your most reliable orthopedic implant partner.
Two decades of focused orthopedic manufacturing gives us unmatched insight into clinical requirements, regulatory landscapes, and supply-chain efficiency.
Every implant batch is validated using precision instruments imported from Germany — including CMM dimensional inspection, tensile testing, and surface roughness analysis.
Our quality management system is certified to both ISO 9001 and ISO 13485 standards. All products carry CE marking, enabling direct entry into EU and many global markets.
Active business relationships in 50+ countries across the Middle East, Africa, Southeast Asia, Eastern Europe, Australia, North America, and South America.
Our dedicated R&D team holds 10+ technical patents and continuously develops new implant systems to meet evolving surgical techniques and market demands.
We pay much attention to improving product quality and perfecting our service. From pre-sale consultation to after-sale technical support, you are never alone.
Our 32,068 m² Hangzhou campus integrates the full production lifecycle — from raw titanium billets to sterile-packaged finished implants — under one roof.
Equipped with 5-axis CNC machining centers capable of producing complex implant geometries with tolerances as tight as ±0.01 mm, ensuring perfect fit and function in surgical applications.
In-house CMM (Coordinate Measuring Machine) inspection, hardness testing, tensile/fatigue testing, and surface finish analysis — all using German-imported equipment for traceable results.
Dedicated passivation, anodizing (for titanium), and electropolishing lines ensure biocompatible, corrosion-resistant implant surfaces meeting ASTM F86 and ISO 10993 standards.
ISO Class 8 cleanroom packaging environment with EO (ethylene oxide) sterilization capability, meeting EU MDR and FDA sterile packaging requirements for export markets.
Every implant is traceable from certified titanium alloy (Ti-6Al-4V ELI, ASTM F136) or 316L stainless steel billet through to finished product, with full material certificates on file.
A dedicated 800 m² R&D center houses our engineering team of 15+ specialists focused on finite element analysis (FEA), biomechanical testing, and new product development cycles.
Enterprise has passed ISO 9001 and ISO 13485 system certification; products have passed CE certification. Our compliance framework ensures your regulatory submissions are supported by robust technical documentation.
| Certification / Standard | Scope | Market Applicability | Status |
|---|---|---|---|
| ISO 13485:2016 | Medical Device Quality Management System — design, manufacture, and distribution of orthopedic implants and instruments | Global (EU, Asia, Americas) | Active |
| ISO 9001:2015 | General Quality Management System — all manufacturing and operational processes | Global | Active |
| CE Marking (MDD/MDR) | Orthopedic implants and surgical instruments — conformity with EU Medical Device Regulation | European Union & EEA | Active |
| ASTM F136 / F138 | Material standard compliance for Ti-6Al-4V ELI and 316L stainless steel implant-grade materials | USA, Global | Compliant |
| ISO 10993 | Biological evaluation of medical devices — biocompatibility testing for all implant materials | Global | Compliant |
| 10+ Technical Patents | Proprietary implant designs, surgical instrument mechanisms, and manufacturing process innovations | China (CNIPA) / PCT | Granted |
Self-produced and self sold, supporting customization — DTRX is a preferred OEM partner for medical device distributors, hospital procurement teams, and orthopedic brands worldwide. We take your concept from drawing to certified finished product.
Share your clinical needs, target market, and regulatory requirements. Our engineers conduct a feasibility assessment within 48 hours.
3D CAD design, FEA simulation, and rapid prototyping using CNC machining. First-article samples delivered for your evaluation and approval.
Mechanical testing, biocompatibility evaluation, and preparation of technical files to support your CE, FDA, or local regulatory registration.
Mass production under ISO 13485 QMS with your private label, custom packaging, and sterile barrier system. On-time delivery worldwide.
From emerging markets to established healthcare systems, DTRX has successfully delivered orthopedic implant programs for distributors, government tenders, and hospital groups worldwide.
Saudi Arabia · Ministry of Health Supply Program
Indonesia · Private Label Distributor
Poland · Multi-hospital Orthopedic Procurement
We do business with customers from over 50 countries. Our regional expertise ensures compliant, on-time delivery tailored to each market's regulatory and logistical requirements.
Our "care first" philosophy translates into long-term partnerships. Here is what our global clients say about working with DTRX Medical Technology.
"DTRX has been our exclusive spinal implant supplier for 4 years. Their ISO 13485 documentation is impeccable — it made our local SFDA registration straightforward. Product quality is consistently excellent, and their technical team responds within hours."
"We placed our first OEM order with DTRX for 3,000 intramedullary nails under our brand. The prototyping process was smooth, the first-article samples passed our internal testing perfectly, and delivery was on schedule. Very professional team."
"As a hospital procurement manager, I appreciate DTRX's traceability system. Every implant comes with a full material certificate and batch record. Their CE-marked bone plates have been used in over 200 surgeries at our hospital with zero implant-related complications."
We keep innovation and have full passion to be better. We are looking forward to having business with you. Let us discuss your orthopedic implant requirements today — whether standard catalogue products, OEM manufacturing, or regulatory support.
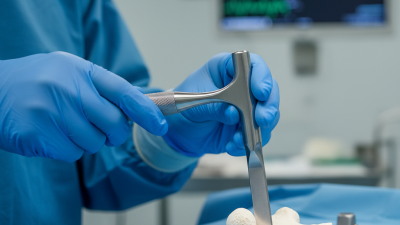
The "Orthopedic Osteotome" plays a crucial role in surgical procedures. This specialized instrument allows surgeons to cut and reshape bone with
Author: Aria Date: 2026-03-30
Learn More
At the 2026 Canton Fair, one of the standout innovations will be Vertebral Body Replacement solutions. This industry is pivotal in addressing spinal
Author: Isabella Date: 2026-03-27
Learn More
At the upcoming Canton Fair 2026, the focus on medical devices will bring significant attention to innovations such as the Proximal Humeral Locking
Author: Henry Date: 2026-03-25
Learn More